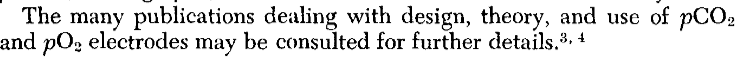Clark and Lyons (1962): Often Cited, Rarely Read
On the quiet foundations of electrochemical biosensing
The published scientific literature is a noisy, chaotic place… where most research goes to die. Paywalls, subscriptions, expired credentials, broken links, university emails we no longer have access to, and two-factor authentications makes visiting a painful experience. Some papers are good, some papers are bad, most are useless.
However, visiting the literature doesn’t always have to be a painful experience and some papers really are worth reading. Remember, the reason you got involved in science and engineering in the first place is because you found it interesting!
So today instead of just skimming through a paper looking for key words in the abstract and conclusion, let’s take a little more time to appreciate the science and scientists who came before us.
‘‘If I have seen further it is by standing on the shoulders of giants.” Is a quote often attributed to Sir. Isaac Newton (which also inspired the name for this section of my Substack). As scientists we like to think of this quote in terms of what we are discovering now, because it is a consequence of all the great science that has come before us.
So in the field of electrochemical biosensing… who are the giants upon whose shoulders we are now standing?
The birth of the biosensing field is often associated with the coining of the term “enzyme electrode” in 1962 by Leland Clark Jr. and Champ Lyons.
Clark was a biochemist and electrochemist best known for inventing the Clark oxygen electrode, the first practical electrochemical sensor capable of measuring dissolved oxygen in blood. His work laid the physical and electrochemical foundations for translating chemical concentrations into reliable electrical signals. Lyons, a cardiovascular surgeon, brought the clinical perspective, ensuring that these devices addressed real physiological and surgical needs rather than remaining purely laboratory instruments.
Together, their collaboration demonstrated a radical idea for the time: biological specificity could be integrated directly onto an electrochemical transducer.
The original paper ‘‘Electrode Systems for Continuous Monitoring in Cardiovascular Surgery’’ has been cited thousands of times and you have definitely seen it referenced in many modern biosensor papers.
You might even think you can imagine what was discussed in the paper without reading it. However, you’d be wrong!
It is also, somehow, still paywalled - but I have attached the PDF below.
Unburdened by the need to grab a reader’s attention quickly, many aspects of the paper also provide insights into how we should be writing our scientific papers, i.e. where the reader is treated as someone to be taught, not dazzled. Many modern papers are written backwards. A speculative idea is framed as the centrepiece, with applications stretched around it - or some surprise findings are suggested as the initial hypotheses. But Clark and Lyons do the opposite. They build a solid, clinically validated measurement system first, and only then gesture toward future specificity.
Overall, the paper is concerned with quantifying the partial pressure of blood oxygen (pO2) and carbon dioxide (pCO2). These values are directly proportional to their respective concentrations in blood, information which is essential for real-time monitoring of a patient's respiratory and metabolic status during surgery.
Surprisingly, the enzyme electrode is not the subject of this paper - it is a footnote. Clark and Lyons weren’t yet aware of the impact this idea would have, and they make no attempt to elevate it beyond a cautious suggestion.
The paper opens with a calm, almost understated tone:
I particularly like this wording: ‘‘Instrument capable of…. indicating the chemical composition of blood have proved useful…’’. They aren’t immediately throwing their best results at you and celebrating how successful their research was.
They go on to discuss what’s currently happening in the field (which is normal) but what I appreciate is that they mention what is working well and what isn’t. This helps stop the proliferation of bad practices and gives readers a high level overview, allowing them to think more critically and come up with more effective research ideas for themselves.
From my experience, if today’s papers shared more insights into the challenges and setbacks encountered when carrying out experimentation - along with the practical tips and tricks individuals pick up from experience - then a lot more research could be reproduced, expanded, and improved upon. This type of knowledge is being passed on less and less!
Clark and Lyons also provide further references for understanding the design, theory, and use of pCO2 and pO2 electrodes. This allows people with less knowledge the opportunity to find out more if they want to, but also keeps the text concise and relevant - making it easier to read.
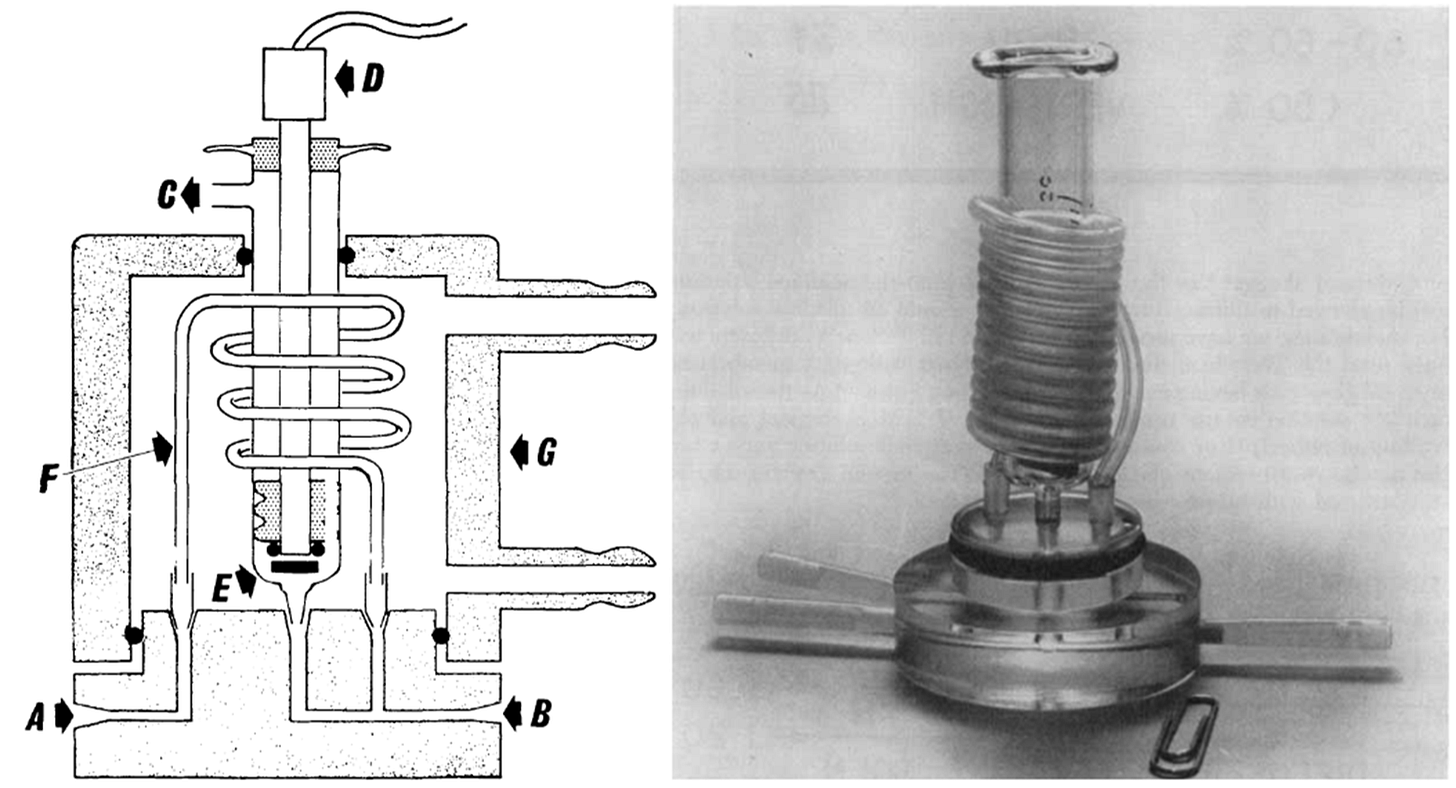
The paper then continues with a discussion around some of the principles of continuously monitoring blood. The black and white schematics at first glance seem primitive but you will quickly notice that they are informative and easy to understand.
Blood from the patient is mixed (continuously) with haemolysing solution (saponin) which lyses red blood cells so the haemoglobin is fully exposed. At sensor A, ferricyanide is introduced which converts oxyhaemoglobin to methaemoglobin, releasing all bound oxygen for electrochemical measurements.
This step overcame a fundamental limitation of electrochemical oxygen electrodes of the time, which responded only to dissolved oxygen, by chemically forcing haemoglobin-bound oxygen into solution.
The liberated oxygen then diffuses through an oxygen-permeable membrane to a platinum cathode held at a constant polarising potential, where it is reduced under diffusion-limited conditions. The resulting steady-state reduction current is proportional to the total oxygen content of the blood sample.
I like how this section of the paper also mentions some practical guidance for carrying out these measurements. This would allows others to expand on this work more effectively - which should be in the back of all good scientists' minds when reading and writing papers.
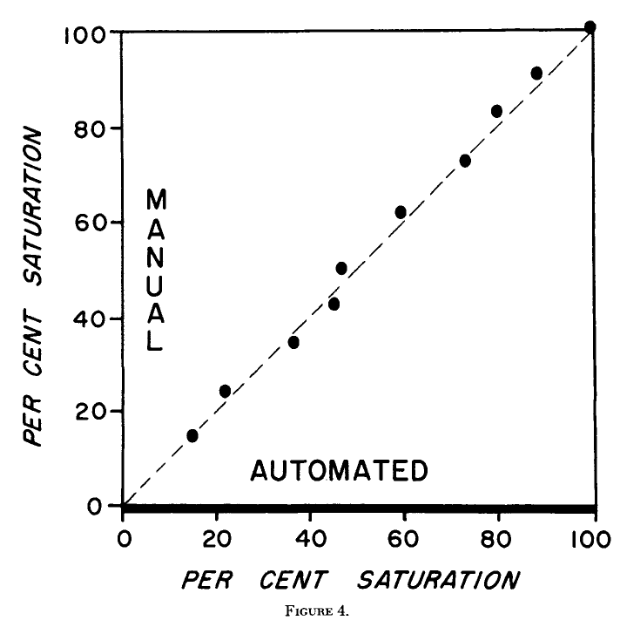
The results obtained using this electrochemical system were compared with those from the Van Slyke procedure, which at the time represented the accepted reference method for determining blood oxygen content.
The Van Slyke procedure was a static, batch-based manometric technique in which discrete blood samples were treated under vacuum to liberate bound gases and the evolved gas volume measured. While highly accurate, the method was labour-intensive and discontinuous, making it unsuitable for continuous measurements. Agreement between the two methods was therefore used to validate that chemically liberated haemoglobin-bound oxygen could be measured accurately using an amperometric oxygen electrode.
Clark and Lyons were modest and clear in presenting their findings, explicitly highlighting the conditions under which performance diminished during real measurements:
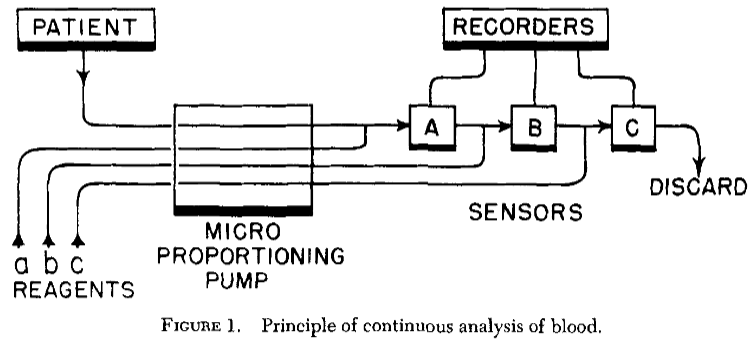
Rather than relying on traditional static cuvette measurements, Clark and Lyons go on to outline a continuous-flow, thermostated system in which anaerobically haemolysed blood is circulated past a microcathode oxygen electrode.
This design enables continuous measurement of both pO2 and oxygen content, simplifies calibration, and improves practical usability without increasing circuit complexity.
What I think is useful in this section is the inclusion of photographs of the equipment set up. They give the reader a concrete sense of what the apparatus actually looked like, rather than leaving the setup abstract or diagrammatic. This helps anchor the method in physical reality, showing how the system was assembled and operated -rather than how it was ideally represented.
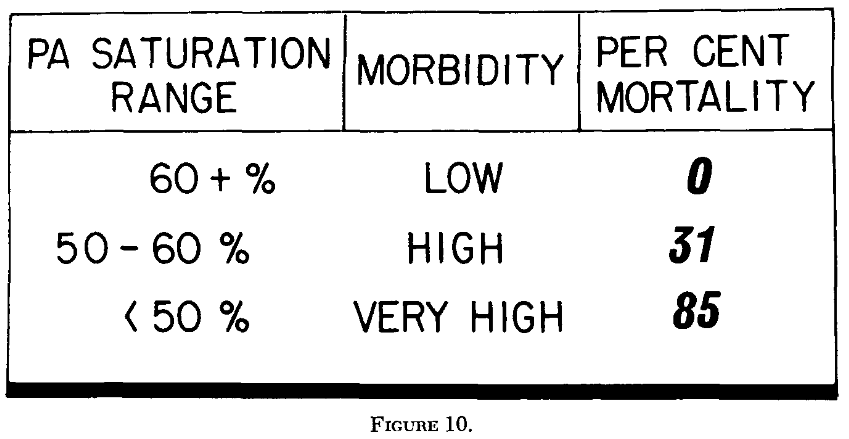
What follows in the paper reads less like a conventional results section and more like an argument for why continuous chemical measurement matters at all. Clark and Lyons show that mixed venous oxygen saturation, when recorded continuously, correlates strongly with postoperative cardiovascular status. Patients with higher saturations consistently survived with routine care, while progressively lower saturations were associated with increasing morbidity and mortality.
Their use of language when it comes to trying to save patients who had low oxygen saturation levels during surgery reads like a novel from a bygone era, yet still effectively communicates the real life impact of their work.
It’s interesting that they don’t present these values as diagnostic thresholds. Clark and Lyons are careful to frame mixed venous oxygen saturation as a continuously evolving physiological signal rather than a single laboratory value. The emphasis is not on prediction, but on interpretation, using real-time chemical information to guide clinical judgement as conditions change.
Clark and Lyons then describe a continuous-flow, thermostated system in which anaerobically haemolysed blood is circulated past a microcathode oxygen electrode.
The design allows both oxygen tension and total oxygen content to be measured within the same system, while maintaining strict control over temperature, mixing, and exposure to ambient oxygen.
Again the schematics are highly informative, showing clearly how fluid handling, reagent addition, and electrochemical sensing are integrated into a single measurement environment. The focus is not on miniaturisation or elegance, but on robustness and interpretability.
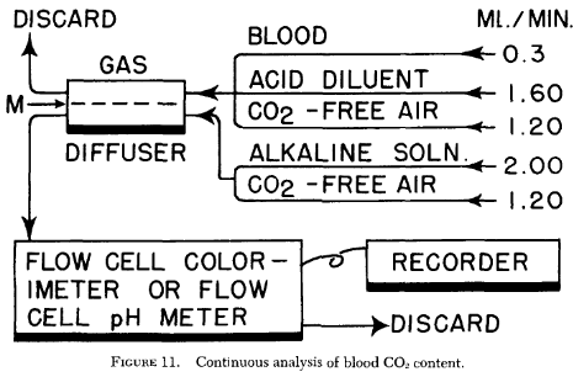
Carbon dioxide measurements are incorporated into the same continuous-flow framework, but are treated very differently. Clark and Lyons do not present CO₂ analysis as a new sensing principle. Instead, established acidification and diffusion-based methods are integrated to demonstrate that multiple blood gases can be monitored continuously using the same system architecture.
The concluding sections of the papers are particularly nice to read and I encourage you to take the time to absorb them. Initially, Clark and Lyons move away from external measurement systems and consider how electrochemical signals behave when the electrode itself is placed directly within the bloodstream.
It’s refreshing to read about how fundamental electrochemical behaviour (changes in potential, redox state, and oxygen tension) can be used pragmatically as physiological indicators, rather than being forced into the kind of tightly parameterised quantitative frameworks that are more common today.
Clark and Lyons make this particularly concrete when they note that:
Reading this now, it’s hard not to feel that this way of thinking still has something to offer. Science has become more complicated in many ways, but that doesn’t mean the best path forward is always to make our models more complex as well.
This leads to my personal favourite section of the paper, one that all biosensor developers should read at least once, the moment an idea is articulated clearly enough that, in hindsight, an entire field begins to feel inevitable.
The idea is simple but profound: rather than forcing chemical specificity into the electrode itself, specificity is provided biologically, upstream of the transducer.
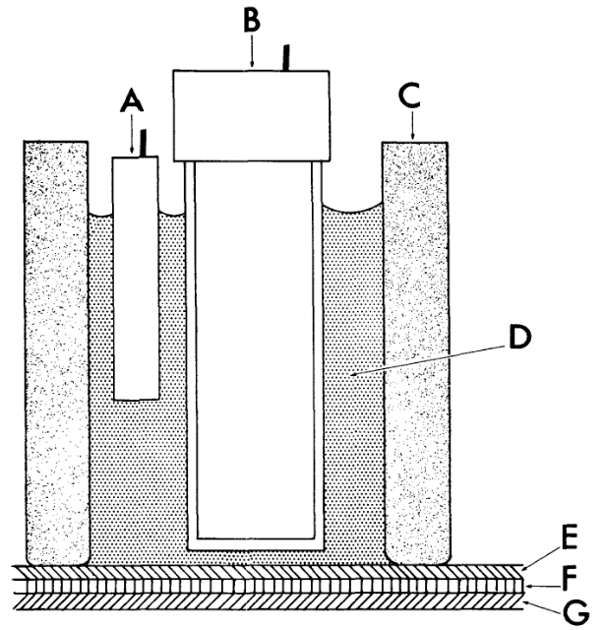
The basic idea is captured schematically:
The electrochemical cell consists of a reference electrode (A) and a sensing electrode (B) housed within a cylindrical body (C) filled with a suitable electrolyte. The end of the cylinder is covered by a multilayer membrane structure. A thin layer of concentrated enzyme (F) is trapped between two membranes (E) and (G). The outer membrane (G) controls transport of analyte from the sample, while the inner membrane (E) electrically and ionically isolates the electrode from the bulk solution. The sensing electrode may be a pH, oxygen, conductivity, or other electrochemical sensor, and responds not to the analyte directly but to products of the enzymatic reaction (such as changes in pH or oxygen concentration). The membranes are held tightly against the electrode surface, forming thin layers that enable rapid response. The entire assembly is intended to be thermostated.
What’s striking is how complete this idea already is. Recognition, transport, and transduction are cleanly separated, yet tightly coupled. The electrode itself remains deliberately generic, responding only to fundamental physical changes, while chemical specificity is imposed biologically and controlled spatially by membrane design. This is, in essence, the modern electrochemical biosensor architecture laid out in plain view.
Clark and Lyons describe this cautiously, even modestly, explicitly noting that the approach is still exploratory. Within little more than a year, enzyme electrodes based on this exact principle would begin to appear, laying the groundwork for what would later become the field of electrochemical biosensing.
In typical Clark and Lyons fashion, they don’t dwell on this idea for long. Having quietly sketched out what would later become the foundation of electrochemical biosensing, the paper simply moves on.
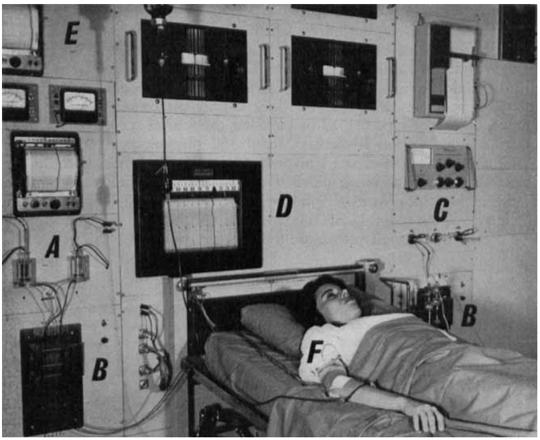
The paper closes with photographs of a working prototype, an “analytical wall” designed to integrate pumps, sensors, recorders, and indicators directly into the clinical environment.
I find there is something quietly reassuring about this ending to the paper. It doesn’t end with claims about the future, but with evidence of work done, systems built, and measurements made.
It’s a reminder that science usually moves forward through patient, careful effort - the kind that rarely announces itself as important at the time.
Thanks for reading along, and thank you for supporting Electrochemical Insights.